- Computational Chemistry
Current Research Studies
- Mechanistic studies in Biochemistry
- Theoretical Studies on the biosyntheses of biological active compounds
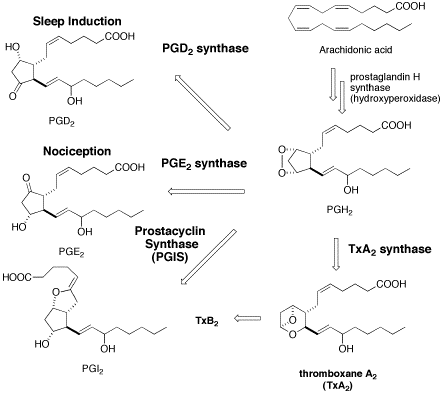
Prostanoid biosyntheses with the aid of quantum mechanical calculations. PGIS and TXAS are in cytochrome P450 superfamily. (TXAS: Chem. Asian J. 2008, PGIS: Chem. Eur J. 2009 ) Role of water molecules in the proton-coupled electron transfer (PCET) process in an unusual cytochrome P450 caralyzed reaction (J. Mol. Graph. Model. 2014).
- We have also examined bilin reductase to compare neutron crystallographic structures (ChemPhysChem. 2018) NEW!
- Our review "Mechanistic Insights into Prostanoid Transformations Catalyzed by Cytochrome P450. Prostacyclin and Thromboxane Biosynthesis " is published in " Advances in Medicine and Biology, Volume 15, Chapter 8, pp. 231-253. "Ed. by L. V. Berhardt fromNova Science Publishers). Download is available from here.(The printing of this material is not available.)
- Toward the studies for understanding of hematopoietic PGDS (J. Inorg. Biochem. 2010)
- PGDS/PGES model reaction mechanisms(Theor. Chem. Acc. 2011 )
- Docking study between biological active compounds and proteins
- Interaction between metal species and biomolecules
We investigated the interaction of group 12 metal ions (Hg2+, Cd2+, Zn2+) with a cysteine/deprotonated cysteine molecule with DFT and high-level ab initio calculations. Water microsolvation approach is necessary to predict higher affinity of Hg2+ than Zn2+ with a cysteine molecule in aqueous solution. Unexpected results appeared in this theoretical studies. (Theor. Chem. Acc. 2011 )
- Theoretical Studies on the biosyntheses of biological active compounds
- Mechanical Studies on Organic Reactions
- metal-mediated reactions
Tungsten-mediated cycloaddition (Chem. Eur. J. 2009), Os-catalyzed hydroxylation (J. Am. Chem. Soc. 2012), Ni-, Rh- and Pd- catalyzed C-O and C-CN bond activation reactions (Bull. Chem. Soc. Jpn, 2014,Dalton Trans 2014, J. Am. Chem. Soc. 2017) NEW!
- Stereoselective catalytic reactions
With aid of IMOMM studies, we are studying the origin of stereoselectivity of enantioselective and diastereoselective reactions. Recently, we found the Rh(I)-BINAP catalyzed hydrogenation with enamides goes through the major Rh(I)(BINAP)(enamide) complex with "lock-and-key" principle as opposed to the other Rh(I)-catalyzed hydrogenation. (Chem. Asian J. 2006)
- Pericyclic reactions
Turquoselectivity of oxetene ring opening to afford unusual high selectivity of alkenes in the reaction of a lithium ynolate with carbonyl compounds. (Review: Synlett 2009; J. Am. Chem. Soc. 2002, Chem. Eur. J. 2006, Org. Lett. 2004, J. Am. Chem. Soc. 2009 , Synlett 2013, and so on)
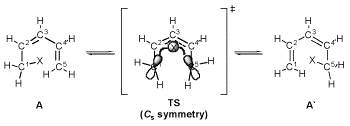
In the [1,5]-sigmatropic hydrogen shift from 1,3-pentadiene, the primary kinetic isotope effects (kH/kD = 12.2 at 298 K) has been already determined for fourty years. Nevertheless, the reason has not been still clarified. With conventional HF and MP2 calculations with Born-Oppenheimer approximation, the KIE values are too small. We have had to use the semiclassical treatment for the tunneling corrections. Recently, with the aid of the MC_MO method developed by Prof. Tachikawa, which treats a hydrogen nuclei as a wavefunction, the KIE value is found to be much closer to the experimental values (8.28 at 298 K). (J. Phys. Chem. A. 2007)
- Theoretical and experimental studies on organometallic reactions
We have examined the reaction mechanisms of several important organocuprate reactions (J. Am. Chem. Soc. 2000; Organometallics 2004; Angew. Chem. Int. Ed. 2005, and so on). Impotrance of multi-point hydrogen bonding interactions and dispersive attractions in Cu(I)-catalyzed enantioselective C-C bond formation (Chem. Eur. J. 2013 and Chem. Sci. 2018)NEW!.